IMPORTANT SAFETY INFORMATION
Cardiomyopathy. A decrease in left ventricular ejection fraction (LVEF) ≥10% below baseline occurred in 23% of 74 pediatric patients who received Koselugo in SPRINT. Four percent of patients experienced decreased LVEF below the institutional lower limit of normal (LLN). Grade 3 decreased LVEF occurred in one patient and resulted in dose reduction. All patients with decreased LVEF were asymptomatic and identified during routine echocardiography. Decreased LVEF resolved in 71% of these patients. Decreased LVEF resulting in permanent discontinuation of Koselugo occurred in a pediatric population with NF1 in an expanded access program. The safety of Koselugo has not been established in patients with a history of impaired LVEF or a baseline ejection fraction that is below the institutional LLN.
Assess ejection fraction by echocardiogram prior to initiating treatment, every 3 months during the first year of treatment, every 6 months thereafter, and as clinically indicated. Withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction. In patients who interrupt Koselugo for decreased LVEF, obtain an echocardiogram or a cardiac MRI every 3 to 6 weeks. Upon resolution of decreased LVEF, obtain an echocardiogram or a cardiac MRI every 2 to 3 months.
Ocular Toxicity. Blurred vision, photophobia, cataracts, and ocular hypertension occurred in 15% of 74 pediatric patients receiving Koselugo in SPRINT. Blurred vision resulted in dose interruption in 2.7% of patients. Ocular toxicity resolved in 82% of 11 patients. Retinal pigment epithelial detachment (RPED) occurred in the pediatric population during treatment with single agent Koselugo and resulted in permanent discontinuation.
Conduct ophthalmic assessments prior to initiating Koselugo, at regular intervals during treatment, and for new or worsening visual changes. Permanently discontinue Koselugo in patients with retinal vein occlusion (RVO). Withhold Koselugo in patients with RPED, conduct ophthalmic assessments every 3 weeks until resolution, and resume Koselugo at a reduced dose. For other ocular toxicities, withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction.
Gastrointestinal Toxicity. Diarrhea occurred in 77% of 74 pediatric patients who received Koselugo in SPRINT, including Grade 3 in 15% of patients. Diarrhea resulting in permanent discontinuation occurred in 1.4% of patients. Diarrhea resulting in dose interruption or dose reduction occurred in 15% and 1.4% of patients, respectively. The median time to first onset of diarrhea was 17 days, and the median duration was 2 days.
Advise patients to start an anti-diarrheal agent (eg, loperamide) and to increase fluid intake immediately after the first episode of diarrhea. Withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction.
Skin Toxicity. Rash occurred in 91% of 74 pediatric patients who received Koselugo in SPRINT. The most frequent rashes included dermatitis acneiform (54%), maculopapular rash (39%), and eczema (28%). Grade 3 rash occurred in 8% of patients. Rash resulted in dose interruption in 11% of patients and dose reduction in 4% of patients. Monitor for severe skin rashes. Withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction.
Increased Creatine Phosphokinase (CPK). Increased CPK occurred in 76% of 74 pediatric patients who received Koselugo in SPRINT, including Grade 3 or 4 in 9% of patients. Increased CPK resulted in dose reduction in 7% of patients. Increased CPK concurrent with myalgia occurred in 8% of patients, including one patient who permanently discontinued Koselugo for myalgia.
Obtain serum CPK prior to initiating Koselugo, periodically during treatment, and as clinically indicated. If increased CPK occurs, evaluate patients for rhabdomyolysis or other causes. Withhold, reduce dose, or permanently discontinue Koselugo based on severity of adverse reaction.
Increased Levels of Vitamin E and Risk of Bleeding. Koselugo capsules contain vitamin E (10 mg capsules contain 32 mg vitamin E as the excipient, D-alpha-tocopheryl polyethylene glycol 1000 succinate [TPGS], while Koselugo 25 mg capsules contain 36 mg vitamin E as TPGS). Vitamin E can inhibit platelet aggregation and antagonize vitamin K-dependent clotting factors. Daily vitamin E intake that exceeds the recommended or safe limits may increase the risk of bleeding. Supplemental vitamin E is not recommended if daily vitamin E intake (including the amount of vitamin E in Koselugo and supplement) will exceed the recommended or safe limits.
An increased risk of bleeding may occur in patients who are coadministered vitamin-K antagonists or anti-platelet antagonists with Koselugo. Monitor for bleeding in these patients and increase international normalized ratio (INR) monitoring in patients taking a vitamin-K antagonist. Perform anticoagulant assessments more frequently and adjust the dose of vitamin K antagonists or anti-platelet agents as appropriate.
Embryo-Fetal Toxicity. Based on findings from animal studies, Koselugo can cause fetal harm when administered to a pregnant woman. In animal studies, administration of selumetinib to mice during organogenesis caused reduced fetal weight, adverse structural defects, and effects on embryo-fetal survival at approximate exposures >5 times the human exposure at the clinical dose of 25 mg/m2 twice daily. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Koselugo and for 1 week after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with Koselugo and for 1 week after the last dose.
Breastfeeding. Due to the potential for adverse reactions in a breastfed child, advise women not to breastfeed during treatment with Koselugo and for 1 week after the last dose.
Concomitant use of Koselugo with a strong or moderate CYP3A4 inhibitor or fluconazole increased selumetinib plasma concentrations, which may increase the risk of adverse reactions. Avoid coadministration of strong or moderate CYP3A4 inhibitors or fluconazole with Koselugo. If coadministration with strong or moderate CYP3A4 inhibitors or fluconazole cannot be avoided, reduce Koselugo dosage.
Concomitant use of Koselugo with a strong or moderate CYP3A4 inducer decreased selumetinib plasma concentrations, which may reduce Koselugo efficacy. Avoid concomitant use of strong or moderate CYP3A4 inducers with Koselugo.
The most common adverse reactions ≥40% are: vomiting, rash (all), abdominal pain, diarrhea, nausea, dry skin, musculoskeletal pain, fatigue, pyrexia, acneiform rash, stomatitis, headache, paronychia, and pruritus.
INDICATION
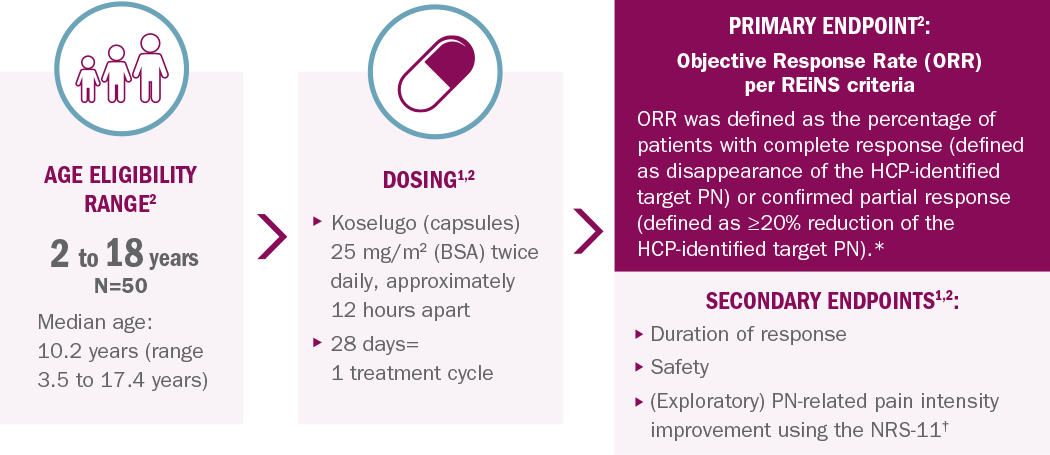
Koselugo is indicated for the treatment of pediatric patients 2 years of age and older with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas (PN).
Please see complete Prescribing Information, including Patient Information.
Please see full Prescribing Information, including Patient Information, by clicking here or at KoselugoHCP.com.
You may report side effects related to AstraZeneca products by clicking here.